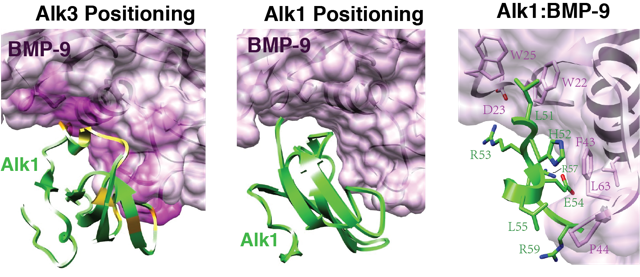
BMP-9 binds and signals through the type I receptor Alk1, but not other Alks, while BMP-2, -4, and -7 bind and signal through Alk3, and the close homolog Alk6, but not Alk1. The results reported herein, which include the determination of the Alk1 structure using NMR and identification of residues important for binding using SPR, show that the b-strand framework of Alk1 is highly similar to Alk3, yet there are significant differences in loops shown previously to be important for binding. The most pronounced difference is in the N-terminal portion of the ß4- ß5 loop, which is structurally ordered and includes a similarly placed but shorter helix in Alk1 compared to Alk3. The altered conformation of the ß4- ß5 loop, and to lesser extent ß1-ß2 loop, cause clashes when Alk1 is positioned onto BMP-9 in the manner that Alk3 is positioned onto BMP-2. This necessitates an alternative manner of binding, which is supported by a model of the BMP-9:Alk1 complex constructed using the program RosettaDock. The model shows that Alk1 is positioned similar to Alk3, but is rotated by 40 degrees. The alternate positioning allows Alk1 to bind BMP-9 through a large hydrophobic interface, consistent with mutational analysis that identified several residues in the central portion of the ß4-ß5 loop that contribute significantly to binding and are non-conservatively substituted relative to the corresponding residues in Alk3.
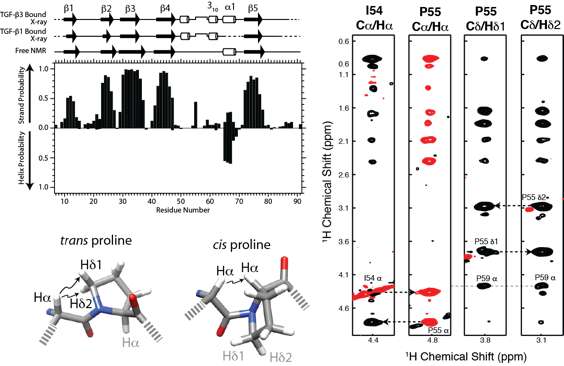
TGF-bs play vital roles regulating cellular growth and differentiation and they signal through a highly restricted subset of receptors, known as TßR-I and TßR-II. TGF-b’s specificity for TßR-I has been proposed to arise from its pre-helix extension, a five residue loop that binds in the cleft between TGF-ß and TßR-II. The structure and backbone dynamics of the unbound form of the TßR-I extracellular domain was determined using NMR to investigate the extension’s role in binding. This showed that the unbound form is highly similar to the bound form, both in terms of the ß-strand framework that defines the three-finger toxin fold and the extension and its characteristic cis Ile54-Pro55 peptide bond. The NMR data further showed that the extension and two flanking 310 helices are rigid on the ns-ps timescale. The functional significance of several residues within the extension was investigated by binding studies and reporter gene assays in cultured epithelial cells. These results demonstrated that the pre-helix extension is essential for binding, with Pro55 and Pro59 each playing a major role. These findings suggest that the pre-helix extension and its flanking prolines evolved to endow the TGF-ß signaling complex with its unique specificity, departing from the ancestral promiscuity of the BMP subfamily, where the binding interface of the type I receptor is highly flexible.
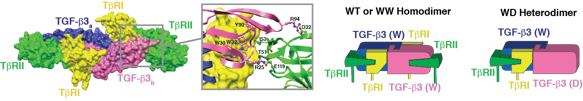
One of the important questions not resolved by the structure of the TGF-ß receptor complex is whether two type I:type II receptor pairs are required for signaling, or whether a single type I:type II receptor pair might suffice. This question was addressed by generating a heterodimeric form of TGF-ß comprised of one wild type monomer and one monomer bearing substitutions to block receptor binding. The receptor binding properties of the purified heterodimer were then studied using highly quantitative surface plasmon resonance (SPR) measurements. The SPR results showed that heterodimeric TGF-ß bound the type II receptor and recruited the type I receptor with affinities indistinguishable from the wild type homodimer, but with one half the stoichiometry. To complete this study, an extensive analysis of the biological activity of the heterodimer was performed, as well as an analysis of the receptor complexes assembled on the cell surface using single-molecule fluorescence. The results very clearly showed that the two type I:type II receptor pairs function independently of one another. This has important consequences for inhibitor design and the lab is leveraging this insight to develop novel protein-based TGF-ß inhibitors.
